Stem cells involved in replenishing human tissues and blood depend on an enzyme known as telomerase to continue working throughout our lives. When telomerase malfunctions, it can lead to both cancer and premature aging conditions. Roughly 90% of cancer cells require inappropriate telomerase activity to survive.
In a groundbreaking new study, an interdisciplinary team of Michigan State University researchers has observed telomerase activity at a single-molecule level with unprecedented precision – expanding our understanding of the vital enzyme and progressing toward better cancer treatments.
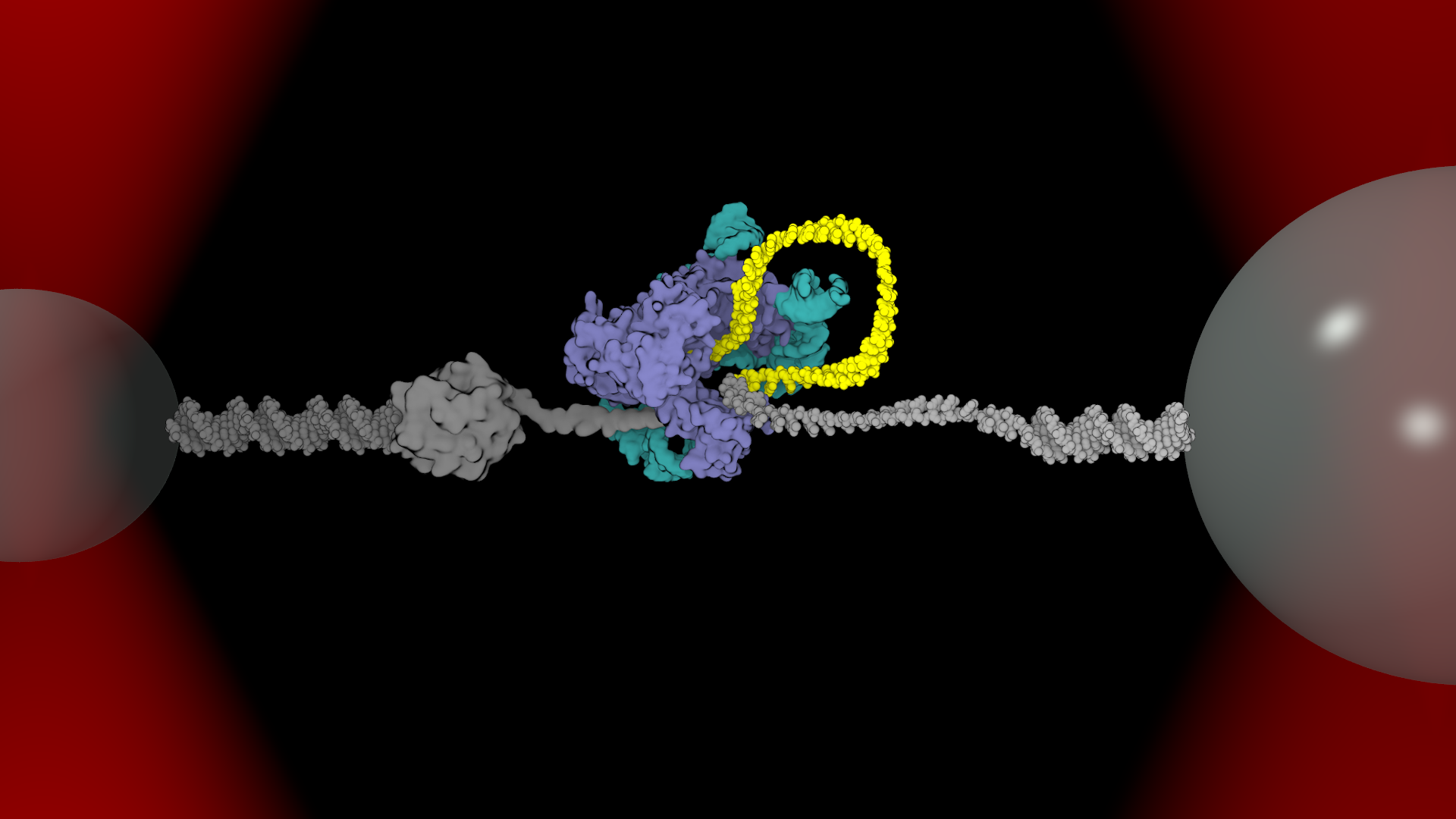
This breakthrough was made possible by a novel investigative procedure and a pair of “optical tweezers,” designed in close collaboration by coauthors Jens Schmidt, assistant professor in the Department of Obstetrics, Gynecology and Reproductive Biology, and Matthew Comstock, assistant professor in the Department of Physics and Astronomy and the Jerry Cowen Endowed Chair of Experimental Physics.
Optical tweezers use powerful lasers to create small forces capable of pushing, pulling and holding microscopic objects, like individual strands of DNA or a telomerase enzyme.
“Our optical tweezer method lets us take the little machines out of the cell, gently hold onto them and watch them go,” Comstock said. “By watching the telomerase work in real time, we can learn how it functions in full detail.”
As stem cells divide over time, the chromosomes gradually decrease in length. Each chromosome end is capped by a telomere – a disposable buffer of repeating DNA sequences.
The telomerase enzyme attaches to the telomere buffer and replaces most of the sequences lost during replication. It was thought that telomerase made progressive extensions in a single step, but scientists could only theorize as to how it stayed in contact and aligned with the right sequence.
What the researchers found was essentially a safety harness anchored to the chromosome in a seemingly specific location.
“In an ideal world we could inhibit telomerase in cancer cells without affecting stem cells,” Schmidt said. “This anchor site is a potential drug target. If we or someone else finds a molecule that interferes with the telomerase anchor site, telomerase would fall off the chromosome end faster, stopping its activity.”
The team hopes their method and discovery will help others in their research.
“It is very important that we also are showing other teams with optical tweezer instruments like ours how to do these experiments,” Comstock said.
For Schmidt and Comstock, this discovery is the realization of a long-term goal – it sets the stage for a wide range of new research opportunities. And most importantly, it brings safer and more effective cancer treatments one step closer.
Both Schmidt and Comstock emphasized the unique partnership between their respective laboratories, with key efforts in the labs from Eric Patrick, research associate, and Joseph Slivka and Bramyn Payne, undergraduate researchers. The pair credits this close relationship as a key factor in the discovery.
The study, “Observation of processive telomerase catalysis using high-resolution optical tweezers,” appears in Nature Chemical Biology.